When it comes to electric vehicle (EV) batteries, the anode material plays a crucial role in how far and how fast your car can go. You might have heard about silicon and graphite anodes, but which one really powers your EV better?
Understanding the difference between silicon and graphite anodes can help you make smarter choices about your next electric car or battery upgrade. Keep reading, because what you learn here could change the way you think about EV performance and battery life.
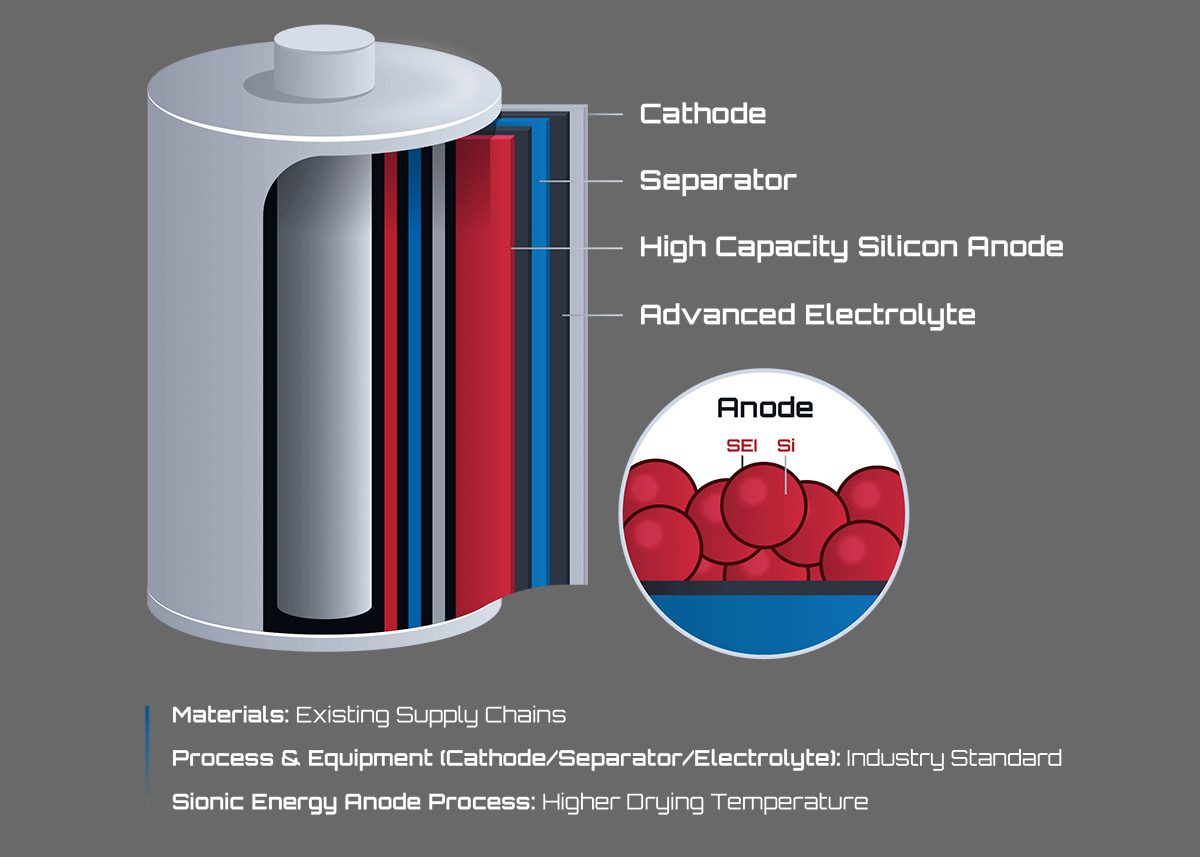
Credit: chargedevs.com
Silicon Anodes Basics
Silicon anodes are becoming popular in electric vehicle (EV) batteries. They offer a new way to store energy. These anodes can hold more lithium ions than traditional materials. This means batteries can last longer and charge faster. Understanding silicon anodes helps us see their role in EV battery technology.
Material Properties
Silicon is a light and abundant element. It can store ten times more lithium than graphite. This high capacity means more energy in smaller spaces. Silicon expands a lot during charging, up to 300%. This expansion can cause cracking and damage. Managing this change is a big challenge for battery makers.
Manufacturing Techniques
Making silicon anodes involves special processes. One method mixes tiny silicon particles with other materials. These particles are then coated onto a copper foil. Another method uses silicon nanowires or thin films. These shapes help reduce cracking from expansion. Careful control during manufacturing improves battery life.
Advantages And Limitations
Silicon anodes store more energy than graphite. This leads to longer driving ranges for EVs. They also support faster charging speeds. But the expansion during charging limits their use. It causes faster wear and shorter battery life. Scientists work to balance these pros and cons for better batteries.
Graphite Anodes Basics
Graphite anodes are key components in electric vehicle (EV) batteries. They store and release lithium ions during charging and discharging. Understanding their basics helps explain why graphite remains popular for anodes. This section breaks down graphite anodes into three parts: material characteristics, production methods, and strengths and weaknesses.
Material Characteristics
Graphite is a form of carbon with a layered structure. Its layers allow lithium ions to move in and out easily. This makes graphite a good conductor of electricity. Graphite is stable at high temperatures. It has a high capacity to store lithium ions. These features make it suitable for battery anodes.
Production Methods
Graphite anodes come from natural or synthetic sources. Natural graphite is mined and purified. Synthetic graphite is made from petroleum coke or coal tar. The material is heated to very high temperatures. This process improves its structure and purity. Both types undergo shaping and coating before use in batteries.
Strengths And Weaknesses
Graphite anodes offer good energy density and long life. They are cost-effective and easy to produce. Graphite works well with current battery designs. Yet, graphite has limits in charging speed and capacity. It can degrade after many cycles. Safety concerns appear if batteries overheat. These factors drive research into alternative materials.
Energy Density Comparison
Energy density is a key factor in electric vehicle (EV) batteries. It measures how much energy a battery stores in a given space. Higher energy density means more power in a smaller, lighter battery. This affects battery size, weight, and cost. Silicon and graphite are two materials used for battery anodes. Each has different energy density properties. Understanding these differences helps see how they impact EV performance.
Capacity Differences
Silicon anodes hold much more lithium than graphite. Silicon can store about 10 times more lithium ions. This leads to a higher capacity for energy storage. Graphite anodes have lower capacity but are very stable. Silicon’s high capacity can increase battery storage size. However, silicon expands a lot during charging, causing stress. This can reduce battery life if not managed well.
Impact On Ev Range
Higher energy density means longer driving range. Batteries with silicon anodes can store more energy in the same space. This can extend EV range without increasing battery size. Graphite anodes offer less range but better cycle life. Using silicon could allow smaller, lighter batteries with good range. EV makers balance these factors to improve vehicle performance.

Credit: www.store-dot.com
Cycle Life And Stability
Cycle life and stability are crucial for EV battery anodes. They affect how long a battery lasts and how well it performs over time. Silicon and graphite anodes behave differently in these areas. Understanding these differences helps in choosing the right material for better battery performance.
Degradation Mechanisms
Silicon anodes expand a lot during charging. This causes cracks and breaks in the material. These cracks reduce battery capacity quickly. Graphite anodes expand less, so they stay stable longer. But graphite also wears down slowly over many cycles. Both materials face chemical changes that degrade their structure.
Longevity In Real-world Use
Graphite anodes typically last longer in everyday driving. They keep stable performance for hundreds of cycles. Silicon anodes can store more energy but lose capacity faster. New designs try to improve silicon’s cycle life. Real-world tests show silicon needs better protection to match graphite’s stability.
Charging Performance
Charging performance is a key factor in electric vehicle batteries. It affects how fast a car can recharge and how safe the process is. The choice between silicon and graphite anodes impacts these aspects. Each material has unique traits that influence charging speed and heat control.
Charge Rates
Graphite anodes offer stable and steady charge rates. They handle charging without much stress on the battery. Silicon anodes can store more energy but face challenges. They swell during charging, which can slow down the process. Engineers work to improve silicon’s ability to charge quickly without damage.
Thermal Management
Managing heat is vital in battery charging. Graphite anodes generate less heat, making cooling easier. Silicon anodes produce more heat because of their volume changes. This heat needs careful control to avoid battery damage. Advanced cooling systems help maintain safe temperatures in silicon-based batteries.
Cost And Scalability
Cost and scalability are key factors in choosing materials for EV battery anodes. The choice affects the final price of batteries and how fast production can grow. Silicon and graphite offer different benefits and challenges in these areas.
Material Costs
Graphite is cheaper and widely available. It has been used for many years in batteries. Silicon costs more because it is less common in battery form. The raw silicon needs special treatment to work well in batteries. This treatment adds to the price. Still, silicon can store more energy, which might save money in the long run.
Manufacturing Challenges
Graphite is easier to use in current battery factories. It fits well with existing equipment. Silicon expands a lot when charging, which can damage the battery. This makes silicon harder to use on a large scale. Manufacturers need new methods to handle silicon safely. These changes take time and money. Scaling up silicon production is still a work in progress.
Safety Considerations
Safety is a key concern in choosing anode materials for EV batteries. Both silicon and graphite have different safety profiles. Understanding these differences helps in making safer battery choices.
Battery safety involves risks like overheating and physical damage. These risks can lead to battery failure or hazards. Let’s explore the main safety aspects of silicon and graphite anodes.
Thermal Runaway Risks
Thermal runaway happens when battery heat builds up uncontrollably. Graphite anodes show stable behavior under normal conditions. Silicon anodes can generate more heat during charge cycles.
More heat means higher chances of thermal runaway. Silicon’s volume changes during charging may increase internal stress. This stress can cause tiny cracks, raising heat risks. Graphite remains more stable, reducing thermal runaway chances.
Mechanical Stability
Mechanical stability is about how well the anode holds up under stress. Graphite expands and contracts less during charging. This property helps graphite keep its shape and function longer.
Silicon anodes can swell up to three times their size. Repeated swelling can break the anode structure. Broken anodes lose contact with the battery, causing performance loss. This instability also raises safety concerns.

Credit: inside.lgensol.com
Future Trends
The future of EV battery anodes will shape the electric vehicle industry. Advances in materials and designs aim to improve battery life and performance. Silicon and graphite each have strengths and limits. New trends try to combine the best of both. These changes will help batteries store more energy and last longer.
Hybrid Anode Designs
Hybrid anodes mix silicon and graphite to boost battery power. Silicon offers high capacity but swells during charging. Graphite is stable but holds less energy. Combining them balances capacity and stability. This design reduces battery wear and improves lifespan. Many companies test hybrid anodes for better EV range. This blend could soon become the standard for EV batteries.
Research And Innovations
Scientists work to solve silicon’s swelling problem. New coatings and materials help control expansion. Nano-structures of silicon improve strength and conductivity. Researchers also explore new binders that hold particles tightly. Innovations focus on making silicon safer and more efficient. Battery labs test these ideas with real EV conditions. Progress in this area promises better, cheaper EV batteries soon.
Frequently Asked Questions
What Are The Main Differences Between Silicon And Graphite Anodes?
Silicon anodes store more lithium, offering higher battery capacity. Graphite anodes are more stable but hold less energy. Silicon can swell, causing durability challenges.
Why Is Silicon Considered For Ev Battery Anodes?
Silicon can increase battery energy by up to 10 times compared to graphite. This means longer driving range for electric vehicles. Its challenge is managing expansion during charging.
How Does Graphite Anode Performance Compare To Silicon?
Graphite offers better cycle life and stability than silicon. It is widely used due to its reliability. But it limits battery capacity growth for EVs.
Can Silicon And Graphite Be Combined In Anodes?
Yes, mixing silicon with graphite improves capacity and stability. This blend helps reduce silicon’s swelling problem. Many EV batteries use this hybrid approach today.
What Impacts Do Silicon Anodes Have On Ev Battery Lifespan?
Silicon anodes may reduce battery lifespan due to expansion and cracking. Careful engineering is needed to maintain long-term performance. Graphite remains better for durability right now.
Conclusion
Silicon and graphite both play key roles in EV battery anodes. Silicon offers higher energy but faces durability challenges. Graphite remains reliable and widely used today. Choosing the right material depends on battery needs and cost. Advances continue to improve both options steadily.
This balance shapes the future of electric vehicle power. Understanding these differences helps make smarter choices. The race for better batteries goes on.
